August 24,2021
Pharma14 adding new EU Parallel Trade database module
Parallel trade is the distribution of an authorized medicinal product from one Member State to another by a pharmaceutical company independent of the marketing-authorization holder.
In the Single Market, parallel traders can buy pharmaceuticals in any EU/EEA country and then under regulated conditions move them to a destination country, repackage them and sell them at a lower cost than the standard local price, thus bringing them into competition with that same identical product sold by the manufacturer or its local licensee.
The turnover of EU parallel imports is estimated to reach approximately €6 billion by 2021.
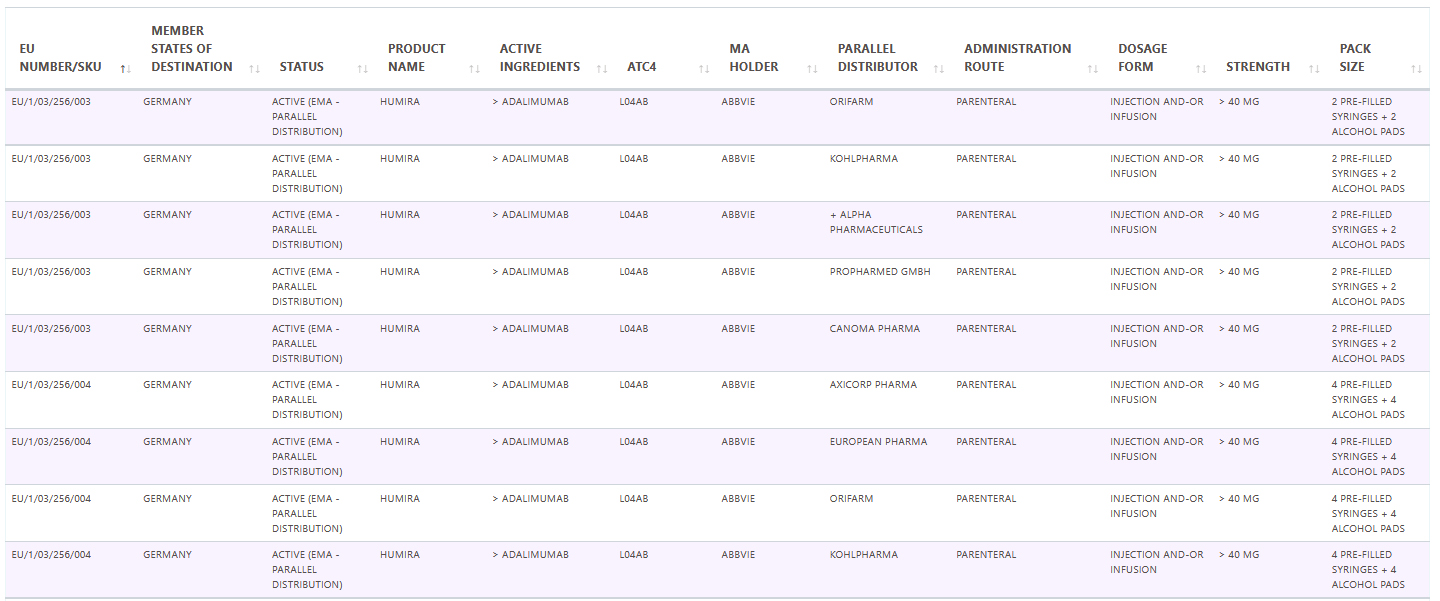
Pharma14 is happy to be adding a dedicated Parallel Trade database and module that will be tracking these medicinal products on a national as well as on a pan European level.
Sample data: January 2021 Adalimumab / Humira in Germany.