April 08,2019
Revlimid first generic versions registered.
Keeping track of first generic medicine registrations of originator molecules is an important part of market intelligence for any pharmaceutical company.
Looking at Celgene’s blockbuster drug Revlimid for example, Pharma14’s proprietary Market Opportunity Locator tool can live track all first generic registrations for Lenalidomide across 39 markets in Europe, North America and more.
The pace of generic registrations can be extremely fast across many markets.
In the case of Lenalidomide we can say that the first generic version registered in Europe was in Latvia by Alvogen in May of this year.
Since this date generic versions of Lenalidomide have been registered in 11 more countries in Europe, primarily by Teva, Accord and Sandoz.
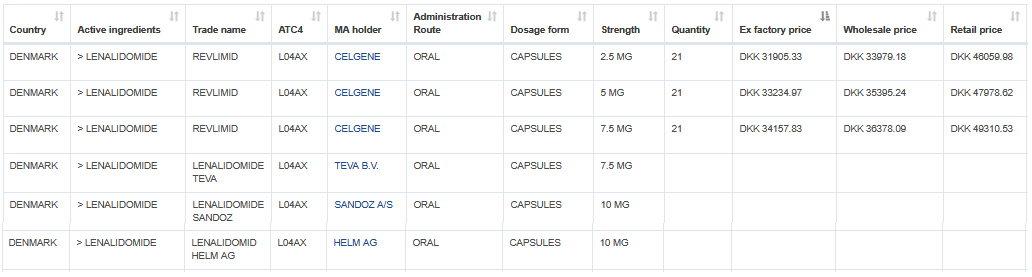
Sample data: Marketed Celgene Revlimid and generic Lenalidomide registrations by Teva, Sandoz and Helm in Denmark