April 07,2019
Silodosin (Rapaflo) analysis. Competitive landscape.
Silodosin (Rapaflo) is prescribed for the treatment of benign prostatic hyperplasia (BPH), or enlarged prostate.
Allergan's Rapaflo lost patent protection in the US on December 1, 2018. It was approved by the FDA in October 2008.
The drug is available in capsule formulation of 4 mg and 8 mg dosages with over $300 million in US and European sales in 2018.
In Europe where the drug was developed by Recordati, the patent expired in 2018 as well (inc.SPC) with clinical data exclusivity until 2020.
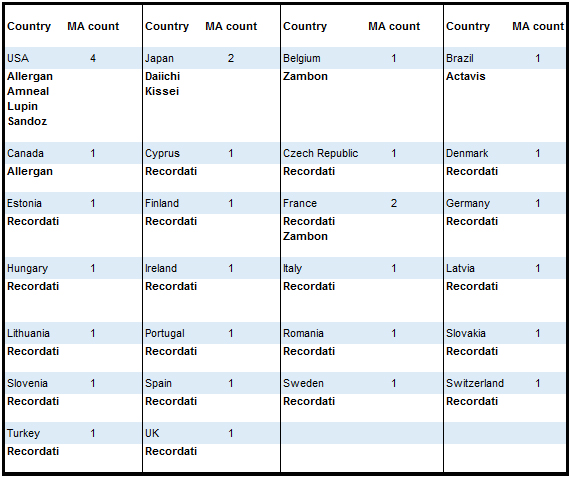
Pharma14’s proprietary Market Opportunity Locator indicates that outside the US market (Lupin, Amneal and Sandoz) no generic marketing authorizations of Rapaflo have been approved.
Pharma14's "Market opportunity locator" is a live proprietary platform that tracks all registrations in 40 countries giving members of our platform direct insight when new generics are registered as well as when they appear on the market.
Sample data: Silodosin. Competitive landscape.